Length: SVL: Males up to 38mm; Females up to 47mm
Weight: up to 7.1 grams
Description
The Hochstetter’s frog is Aotearoa/New Zealand's most widespread, and commonly encountered native frog species. Whilst still in decline they have fared much better than their close relatives (the Hamilton's and Archey's frogs), and still occupy a significant proportion of their former range. This may be attributed to their much more aquatic nature; being almost exclusively associated with small forested catchments or seepages.
Generally, Hochstetter's frogs match the mossy and gravel-like tones of their environment, exhibiting greenish-brown to reddish-brown dorsal (upper) surfaces, punctuated by dark markings and numerous warts. The ventral (lower) surface is yellow-brown. Their forelimbs lack any webbing, whilst their hindlimbs are characterised by partial webbing. As with other Leiopelma frogs, they do not have visible tympana (disc-shaped external eardrums).
Recent work has suggested that Hochstetter's frogs may comprise eleven different taxa (or at the least distinct populations). These are comprised of the Northern Coromandel/Hunuas population (L. hochstetteri), Northland populations (L. "Northland"), Waitākere Ranges populations (L."Waitākere"), Great Barrier Island populations (L. "Great Barrier"), Central/South Coromandel populations (L. "Central/South Coromandel"), Kaimai Ranges populations (L. Kaimai), Otawa population (L. "Otawa"), Western Raukūmara Ranges populations (L. "Western Raukūmara"), Eastern Raukūmara populations (L. "Eastern Raukūmara), Waikato populations (L. "Waikato"), and the Whareorino population (L. "Whareorino).
Hochstetter’s frogs are similar in appearance to our other native frog species but can be differentiated by their squatter build, aquatic nature, and the presence of webbing (although only partial) on their hindlimb. They co-occur with the terrestrial Archey's frog (Leiopelma archeyi) on the Coromandel Peninsula, and in the King Country, but are much larger in size.
Larval stage:
The larvae of the Hochstetter’s frog resemble a small, round fish, lacking fins.
Their colouration changes as they age, but generally they have pink dorsal (upper) surfaces and yellowish-orange ventral (lower) surfaces. Their heads and limbs develop darker tones (various shades of grey) as they mature.
After about 1 month has passed the larvae have fully-developed limbs, with a 6-10mm SVL, and tails ranging from 10-14mm. At this stage, they still retain the larval colouration.
2 months after hatching they are considered froglets, having fully resorbed their tails, and developed the dark-brown or greenish colouration seen in adult frogs.
Life expectancy
Like our other endemic frog species, Hochstetter's frogs are extremely long-lived, reported to reach ages in excess of 30 years (van Winkel et al., 2018). However, unlike our other endemic frog species, the Hochstetter's frog has not had any long-term population studies, and thus accurate estimates of longevity have not been captured. Short-term monitoring has revealed age estimates of at least 18 years (Bell et al., 2023).
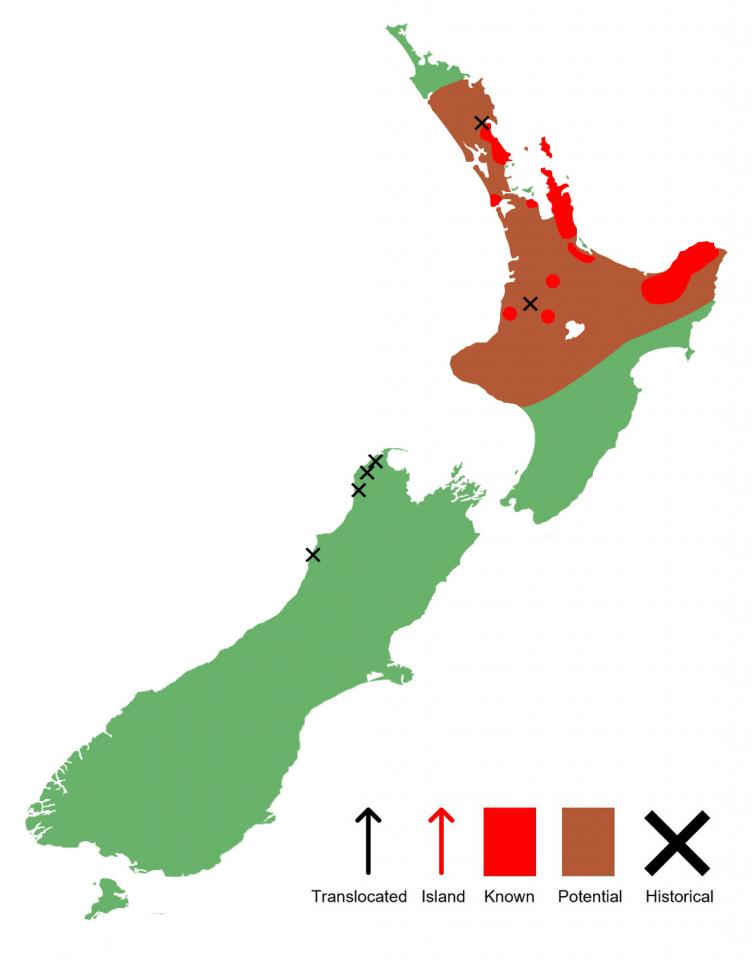
Distribution
Prior to the colonisation of Aotearoa by humans, Hochstetter’s frogs had a much greater distribution, occurring from Punakaiki on the South Island's west coast through to their current northern distribution in Waipu, Northland (Worthy, 1987). Today, they are restricted to the North Island, occurring in discrete populations scattered from the East Cape, across to the King Country, and up to Waipu. An additional population occurs on Aotea/Great Barrier Island within the Hauraki Gulf.
Ecology and habitat
The Hochstetter’s frog is a nocturnal species, which can occasionally be found sitting in shaded areas during daylight hours. Unlike our other extant Leiopelma, Hochstetter's frogs are a semi-aquatic species, typically occurring within the vicinity of small forested streams, with plentiful rocky/woody debris. They are particularly averse to high sediment loads/silt, and as such are often absent from streams with significant disturbance (e.g., pig wallowing, habitat modification, forestry) (Whitaker, 1985; McLennon, 1985; Easton, 2015; Nájera-Hillman, 2009).
Unlike our other Leiopelma species, the young of Hochstetter's frogs are highly reliant on water bodies. This is due to them emerging from the egg at a less-developed stage than the terrestrial species (larvae/tadpole vs. froglet). These larvae exhibit high site fidelity (remaining in one place) until metamorphosis, and possess an aversion to light sources, preferring to bury themselves in debris at the bottom of ponds (Beauchamp et al.). These behaviours are likely related to predator avoidance.
Although they occupy a relatively narrow niche, Hochstetter's frogs can be found in a variety of habitats including; pine forests, mature podocarp/broadleaf forests, regenerating scrubland, seepages in banks, as well as in ditches adjacent to forested areas. Within these habitats, they sit within the mid-trophic level (middle of the food chain), preying on various invertebrates, and being prey for terrestrial predators, and large freshwater fish (e.g., banded kōkopu and tuna (shortfin, and longfin eels)) (Nájera-Hillman, 2009).
Social structure
Largely unknown, however, they appear to be primarily solitary. As with other Leiopelma species they can reach high densities in suitable habitats (Bell, 1994), but are rarely found occupying the same refugia outside of the breeding season. Unlike the exotic frogs that have been introduced to Aotearoa, Leiopelma frogs do not vocalise during the mating season to attract mates, instead, vocalisations are only known to (rarely) occur (squeak or chirp) when they are disturbed.
Some studies have shown that Aotearoa's native frogs utilise chemical signals to remain near their home range, preferring their own biochemistry or familiar biochemistries over those of unrelated populations.
Breeding biology
As with all of our endemic frog species, the Hochstetter’s frog is oviparous (egg-laying), laying either one or two strings of 10-13 small whitish-yellow eggs (4-6mm in diameter), from late September through to May. Prior to laying, male frogs will latch onto females in a process called amplexus, fertilising eggs as the female lays them in a suitable nesting site. There is very limited information on nest preference, but Hochstetter’s frogs appear to select moist sites, sheltered by various debris. In particular, they seem to favour seepages with substantial rock cover, small pools with extensive leaf litter, large invertebrate burrows (New Zealand bush giant dragonfly nymphs(Uropetala carovei)), and underwater cavities (Beauchamp et al., 2010; Bell, 1978).
As is typical of Leiopelma, Hochstetter’s frogs do not have a free-living (feeding) tadpole phase, however, they do have a nidicolous (remain at the nest site for an extended duration) larval phase, unlike our other endemic frogs. These swimming larvae emerge from the gelatinous eggs (which at this stage are about 20-27mm in diameter) around 40 days after being deposited. If they are not already in a suitable water body at this stage, they quickly make their way to the closest one. These larvae emerge with non-digited hindlimbs, but over the next ~52 days slowly form fully digited hind and forelimbs, and reabsorb their tail. By the time metamorphosis is complete the froglets are around 6-11mm SVL. Hochstetter’s frogs reach maturity/breeding age at around 3 years (Beauchamp et al., 2010; Bell, 1978).
As opposed to Archey’s frogs (Leiopelma archeyi), Hochstetter’s frogs do not show any parental care (Beauchamp et al., 2010; Bell, 1978).
Diet
Hochstetter’s frogs are carnivorous, nocturnal predators feeding on a wide range of invertebrate prey. An analysis of the gut contents of wild animals found that their prey consisted of springtails, mites, ants, parasitic wasps, amphipods, and isopods (Shaw et al., 2012), although it is likely that they opportunistically prey on other invertebrates within, and near water bodies.
Disease
As is the case with all of Aotearoa's herpetofauna, the diseases and parasites of our endemic Leiopelma frogs are poorly understood, and incomplete. However, their main disease risk comes in the form of the Amphibian chytrid fungus (Batrachochytrium dendrobatidis (Bd)), a fungal infection that causes a disease called chytridiomycosis. This disease has decimated or caused the extinction of many frog species worldwide. Luckily, our endemic frogs seem to be somewhat immune to it, having peptides within the skin that inhibit the fungus (Shaw et al., 2014; Melzer & Bishop, 2010; Eda et al., 2023).
Whilst, Bd is a major concern for wild populations, some diseases are more prevalent in captive situations. For example, in captivity, Leiopelma frogs are known to develop metabolic bone disease (MBD) and osteofluorosis (accumulation of fluoride in bones) due to improper husbandry (lack of UVB light, and excess fluoride in water bodies) (Shaw, 2012; Shaw et al., 2012).
Additionally, Hochstetter's frogs are a known host for the trematode Dolichosaccus novaezealandiae, a species seemingly restricted to Aotearoa's native frog species (Allison & Blair, 1987), and accordingly, just as threatened.
Conservation strategy
In the most recent threat classification, Hochstetter's frog populations range from 'At Risk - Declining' through to 'Threatened - Nationally Critical'. Of the eleven proposed taxa, seven fall within 'At Risk - Declining', three (L. "Great Barrier", L. "Waikato", and L. "Whareorino") within the 'Threatened - Nationally Vulnerable' category, and one (L. "Otawa") within the ' Threatened - Nationally Critical' category.
In general, amphibians are highly sensitive to environmental change, and Aotearoa's frogs are no different. Hochstetter's frogs are particularly sensitive to their freshwater environments, with any change in chemistry or environmental makeup possibly resulting in major population crashes. Factors such as habitat modification/loss (via logging, mining, or infrastructure development), the release of toxins into waterways (via mining, chemical spills, or wastewater), and pig damage (disturbance of stream edges) are all serious threats to several Hochstetter's frog populations. The disease chytridiomycosis is also a serious threat to our endemic frogs, however, they seem to have some resistance to it.
Additionally, as with the majority of our herpetofauna, they have been negatively impacted by the introduction of mammalian predators, with many populations becoming locally extinct, or highly suppressed in certain regions (Worthy, 1987; Baber et al., 2006; Longson et al., 2017). Introduced frogs have also been implicated as a threat.
Hochstetter's frogs are not currently being managed, however, several populations occur within highly predator-controlled areas (e.g., Maungatautari, Mahakirau, Whareorino Forest, and the kōkako management area in the Hunua Ranges). Although some of these areas often have a focus on protecting key species (e.g., kōkako) they inadvertently have benefits on the wider flora and fauna, as adjacent areas without specific pest control often exhibit smaller, and less robust populations (Crossland et al., 2023).
Interesting notes
The specific name 'hochstetteri' references Ferdinand von Hochstetter (1829-1884), a german-austrian geologist who was responsible for bringing the first specimens to Europe (van Winkel et al., 2018). He was also instrumental in understanding the geological background of Aotearoa's landscapes - mapping Auckland's volcanic fields, as well as the pink and white terraces in the Rotorua District.
The Hochstetter's frog is the sister species to the extinct Markham's frog (Leiopelma markhami), and the aurora frog (Leiopelma auroraensis), with the extant Archey's and Hamilton's frogs being more distant relatives (Worthy, 1987).
As is the case with all of our Leiopelma, Hochstetter's frogs exhibited significant morphological structuring across their historical range, with specimens from the southern extent of their distribution (Nelson) being much larger than northern populations (Worthy, 1987).
During a survey of the northern block of Aotea/Great Barrier Island for Hochstetter's frogs, Tony Whitaker and Graham Hardy came across a pair of frogs that were distinctly different from Hochstetter's frogs in terms of their behaviour. These frogs were found lying in a small pool of a fast-flowing forested stream during the day, and when approached they quickly swam away with a strong double-leg action (Whitaker & Hardy, 1985). Due to this strange behaviour and the habitat that they occupied, it was surmised that this was either an unusual behaviour, or that these frogs represented a cryptic, and as yet undescribed species. These frogs were later referred to as the 'northern Great Barrier swimming frogs' in the literature, but sadly, no new specimens have been found.
References
Allison, B., & Blair, D. (1987). The genus Dolichosaccus (Platyhelminthes: Digenea) from amphibians and reptiles in New Zealand, with a description of Dolichosaccus (Lecithopyge) leiolopismae n. sp. New Zealand journal of zoology, 14(3), 367-374.
Baber, M., Moulton, H., Smuts‐Kennedy, C., Gemmell, N., & Crossland, M. (2006). Discovery and spatial assessment of a Hochstetter's frog (Leiopelma hochstetteri) population found in Maungatautari Scenic Reserve, New Zealand. New Zealand Journal of Zoology, 33(2), 147-156.
Beauchamp, A. J., Lei, P., & Goddard, K. (2010). Hochstetter's frog (Leiopelma hochstetteri) egg, mobile larvae and froglet development. New Zealand Journal of Zoology, 37(2), 167-174.
Bell, B. D. (1978). Observations on the ecology and reproduction of the New Zealand Leiopelmid frogs. Herpetologica, 340-354.
Bell, B. D. (1994). A review of the status of New Zealand Leiopelma species (Anura: Leiopelmatidae), including a summary of demographic studies in Coromandel and on Maud Island. New Zealand journal of zoology, 21(4), 341-349.
Bell, B. D. (2010). The threatened Leiopelmatid frogs of New Zealand: Natural history integrates with conservation. Herpetological Conservation and Biology, 5(3), 515-528.
Bell, B. D. (2016). A review of potential alpine newt (Ichthyosaura alpestris) impacts on native frogs in New Zealand. Journal of the Royal Society of New Zealand, 46(3-4), 214-231.
Bell, B. D., Shirley, A., & Pledger, S. (2023). Post-metamorphic body growth and remarkable longevity in Archey's frog and Hamilton's frog in New Zealand. New Zealand Journal of Ecology, 47(2), 3528.
Bell, B. D., & Wassersug, R. J. (2003). Anatomical features of Leiopelma embryos and larvae: implications for anuran evolution. Journal of Morphology, 256(2), 160-170.
Benno Meyer-Rochow, V., & Pehlemann, F. W. (1990). Retinal organisation in the native New Zealand frogs Leiopelma archeyi, L. hamiltoni, and L. hochstetteri (Amphibia: Anura; Leiopelmatidae). Journal of the Royal Society of New Zealand, 20(4), 349-366.
Bishop, P. J., Daglish, L. A., Haigh, A., Marshall, L. J., Tocher, M., & McKenzie, K. L. (2013). Native frog (Leiopelma spp.) recovery plan, 2013-2018: Threatened species recovery plan 63. Department of Conservation, Wellington, New Zealand.
Bowsher, J. H. (2000). Intraspecific genetic variation in New Zealand's endemic frog, Leiopelma hochstetteri: a thesis submitted in fulfilment of the requirements for the Degree of Master of Science in the University of Canterbury.
Burns, R. J., Bell, B. D., Haigh, A., Bishop, P., Easton, L., Wren, S., Germano, J., Hitchmough, R. A., Rolfe, J. R., & Makan, T. (2018). Conservation status of New Zealand amphibians, 2017. New Zealand Threat Classification Series 25: Department of Conservation, Wellington. 7 p.
Burns, R. J., Armstrong, D. P., Bell, B. D., Haigh, A., Germano, J., Rawlence, N. J., Thurley, T., Hitchmough, R. A., Makan, T., & Michel, P. (2025). Conservation status of amphibians in Aotearoa New Zealand, 2024. New Zealand Threat Classification Series 44: Department of Conservation, Wellington. 19 p.
Carr, L. M., McLenachan, P. A., Waddell, P. J., Gemmell, N. J., & Penny, D. (2015). Analyses of the mitochondrial genome of Leiopelma hochstetteri argues against the full drowning of New Zealand. Journal of Biogeography, 42(6), 1066-1076.
Clay, C., Gleeson, D., Howitt, R., Lawrence, H., Abdelkrim, J., & Gemmell, N. (2010). Characterisation of microsatellite markers for the primitive New Zealand frog, Leiopelma hochstetteri. Conservation Genetics Resources, 2, 301-303.
Crossland, M. R., Kelly, H., Speed, H. J., Holzapfel, S., & MacKenzie, D. I. (2023). Predator control to protect a native bird (North Island kōkako) also benefits Hochstetter's frog. New Zealand Journal of Ecology, 47(2), 3530.
Crossland, M. R., MacKenzie, D. I., & Holzapfel, S. (2005). Assessment of site-occupancy modeling as a technique to monitor Hochstetter’s frog (Leiopelma hochstetteri) populations. DOC Research & Development Series 218. Department of Conservation, Wellington. 23 p.
Daugherty, C. H., Bell, B. D., Adams, M., & Maxson, L. R. (1981). An electrophoretic study of genetic variation in the New Zealand frog genus Leiopelma. New Zealand journal of zoology, 8(4), 543-550.
Daugherty, C. H., Maxson, L. R., & Bell, B. D. (1982). Phylogenetic relationships within the New Zealand frog genus Leiopelma—immunological evidence. New Zealand journal of zoology, 9(2), 239-242.
Easton, L. (2015). Determining the feasibility of a translocation by investigating the ecology and physiology of the threatened Hochstetter's frog (Leiopelma hochstetteri): A thesis submitted for the degree of Masters of Science at the University of Otago, Dunedin, New Zealand.
Easton, L. (2018). Taxonomy and genetic management of New Zealand's Leiopelma frogs (Doctoral dissertation, University of Otago).
Easton, L. J., Rawlence, N. J., Worthy, T. H., Tennyson, A. J., Scofield, R. P., Easton, C. J., Bell, B. D., Whigham, P. A., Dickinson, K. J. M., & Bishop, P. J. (2018). Testing species limits of New Zealand’s leiopelmatid frogs through morphometric analyses. Zoological Journal of the Linnean Society, 183(2), 431-444.
Eda, A. R. A. R., Bishop, P. J., Altobelli, J. T., Godfrey, S. S., & Stanton, J. L. (2023). Screening for Batrachochytrium dendrobatidis in New Zealand native frogs: 20 years on. New Zealand Journal of Ecology, 47(2), 3531.
Egeter, B., Bishop, P. J., & Robertson, B. C. (2011). DNA detects frog predation, University of Otago. FrogLog, 99, 36-37.
Egeter, B., Robertson, B. C., & Bishop, P. J. (2015). A synthesis of direct evidence of predation on amphibians in New Zealand, in the context of global invasion biology. Herpetological Review, 46(4), 512-519.
Egeter, B., Roe, C., Peixoto, S., Puppo, P., Easton, L. J., Pinto, J., Bishop, P. J., & Robertson, B. C. (2019). Using molecular diet analysis to inform invasive species management: A case study of introduced rats consuming endemic New Zealand frogs. Ecology and Evolution, 9(9), 5032-5048.
Eggers, K. E. (1998). Morphology, ecology and development of leiopelmatid frogs (Leiopelma spp.), in Whareorino Forest, New Zealand: a thesis presented in partial fulfilment of the requirements for the degree of Masters of Science in Ecology at Massey University (Doctoral dissertation, Massey University).
Fouquet, A., Ficetola, G. F., Haigh, A., & Gemmell, N. (2010). Using ecological niche modelling to infer past, present and future environmental suitability for Leiopelma hochstetteri, an endangered New Zealand native frog. Biological Conservation, 143(6), 1375-1384.
Fouquet, A., Green, D. M., Waldman, B., Bowsher, J. H., McBride, K. P., & Gemmell, N. J. (2010). Phylogeography of Leiopelma hochstetteri reveals strong genetic structure and suggests new conservation priorities. Conservation Genetics, 11(3), 907-919.
Gemmell, N. J., Bowsher, J. H., & Gomas, K. P. (2003). Genetic affinities of Hochstetter's frog (Leiopelma hochstetteri) populations in the Bay of Plenty: DOC Science Internal Series 141. Department of Conservation, Wellington. 19 p.
Germano, J. M., Earl, R., Tocher, M., Pearce, P., & Christie, J. (2023). The conservation long game: Leiopelma species climate envelopes in New Zealand under a changing climate. New Zealand Journal of Ecology, 47(2), 3535.
Gill, B., & Whitaker, T. (2007). New Zealand frogs and reptiles. Auckland: David Bateman Publishing Ltd.
Green, D. M. (1988). Antipredator behaviour and skin glands in the New Zealand native frogs, genus Leiopelma. New Zealand journal of zoology, 15(1), 39-45.
Green, D. M. (1988). Cytogenetics of the endemic New Zealand frog, Leiopelma hochstetteri: extraordinary supernumerary chromosome variation and a unique sex-chromosome system. Chromosoma, 97(1), 55-70.
Green, D. M. (1994). Genetic and cytogenetic diversity in Hochstetter's frog, Leiopelma hochstetteri, and its importance for conservation management. New Zealand journal of zoology, 21(4), 417-424.
Green, D. M., & Cannatella, D. C. (1993). Phylogenetic significance of the amphicoelous frogs, Ascaphidae and Leiopelmatidae. Ethology Ecology & Evolution, 5(2), 233-245.
Green, D. M., Kezer, J., & Nussbaum, R. A. (1984). Triploidy in Hochstetter's frog, Leiopelma hochstetteri, from New Zealand. New Zealand journal of zoology, 11(4), 457-460.
Green, D. M., Sharbel, T. F., Hitchmough, R. A., & Daugherty, C. H. (1989). Genetic variation in the genus Leiopelma and relationships to other primitive frogs. Journal of Zoological Systematics and Evolutionary Research, 27(1), 65-79.
Green, D. M., & Tessier, C. (1990). Distribution and abundance of Hochstetter's frog, Leiopelma hochstetteri. Journal of the Royal Society of New Zealand, 20(3), 261-268.
Green, D. M., Zeyl, C. W., & Sharbel, T. F. (1993). The evolution of hypervariable sex and supernumerary (B) chromosomes in the relict New Zealand frog, Leiopelma hochstetteri. Journal of evolutionary biology, 6(3), 417-441.
Herbert, S., Melzer, S., Gilbert, J., & Jamieson, H. (2014). Relative abundance and habitat use of Hochstetter’s frog (Leiopelma hochstetteri) in northern Great Barrier Island: a snapshot from 2012. BioGecko, 2, 12-21.
Jewell, T. (2011). A photographic guide to reptiles and amphibians of New Zealand. Auckland: New Holland Publishing.
Johnson, C. E. (2022). A comparison of approaches for estimating Hochstetter's frog (Leiopelma hochstetteri) abundance: a thesis presented in partial fulfilment of the requirements for the degree of Master of Science in Conservation Biology at Massey University, Palmerston North, New Zealand (Doctoral dissertation, Massey University).
Kudeweh, S., Goold, M., & Standley, S. (2011). Hamilton Zoo. FrogLog, 99, 34.
Longson, C. G., Brejaart, R., Baber, M. J., & Babbitt, K. J. (2017). Rapid recovery of a population of the cryptic and evolutionarily distinct Hochstetter's Frog, Leiopelma hochstetteri, in a pest‐free environment. Ecological Management & Restoration, 18(1), 26-31.
McLennan, J. A. (1985). Some observations on Hochstetter's frog in the catchment of the Motu River, East Cape. New Zealand Journal of Ecology, 1-4.
Melzer, S., & Bishop, P. J. (2010). Skin peptide defences of New Zealand frogs against chytridiomycosis. Animal Conservation, 13, 44-52.
Melzer, S., & Bishop, P. J. (2011). Just Juice? Attempting to unravel the secrets of skin secretions in New Zealand's endemic frogs. FrogLog, 99, 37-38.
Melzer, S., Clerens, S., & Bishop, P. J. (2011). Differential polymorphism in cutaneous glands of archaic Leiopelma species. Journal of Morphology, 272(9), 1116-1130.
Melzer, S., Hitchmough, R., van Winkel, D., Wedding, C., Chapman, S., Rixon, M., Moreno, V., & Germano, J. (2022). Conservation Status of Amphibian Species in Tāmaki Makaurau/Auckland. Auckland Council technical report TR2022/4.
Moreno, V., Aguayo, C. A., & Brunton, D. H. (2011). A survey for the amphibian chytrid fungus Batrachochytrium dendrobatidis in New Zealand's endemic Hochstetter's frog (Leiopelma hochstetteri). New Zealand Journal of Zoology, 38(2), 181-184.
Najera-Hillman, E. (2009). Leiopelma hochstetteri Fitzinger 1861 (Anura: Leiopelmatidae) habitat ecology in the Waitakere Ranges, New Zealand: A thesis submitted to the Faculty of Health and Environmental Sciences, Auckland University of Technology in fulfilment of the requirements for the degree of Doctor of Philosophy.
Nájera-Hillman, E. (2012). Evaluation of endemic frog (Leiopelma hochstetteri) habitat through a spatial decision support system. BioGecko, 1, 7-15.
Nájera-Hillman, E., Alfaro, A. C., O’Shea, S., Breen, B., Garret, N., & King, P. (2009). Habitat-use model for the New Zealand endemic frog Leiopelma hochstetteri. Endangered Species Research, 9(1), 23-31.
Nájera-Hillman, E., King, P., Baird, A. C., & Breen, B. B. (2009). Effect of pest–management operations on the abundance and size–frequency distribution of the New Zealand endemic frog Leiopelma hochstetteri. New Zealand Journal of Zoology, 36(4), 389-400.
Newman, D. G. (1996). Native frog (Leiopelma spp.) Recovery plan: Threatened species recovery plan No. 18. Wellington: Department of Conservation.
Newman, D. G., Bell, B. D., Bishop, P. J., Burns, R., Haigh, A., Hitchmough, R. A., & Tocher, M. (2010). Conservation status of New Zealand frogs, 2009. New Zealand Journal of Zoology, 37(2), 121-130.
Newman, D. G., & Towns, D. R. (1985). A survey of the herpetofauna of the northern and southern blocks, Great Barrier Island, New Zealand. Journal of the Royal Society of New Zealand, 15(3), 279-287.
Ohmer, M., & Bishop, P. J. (2011). Susceptibility to chytridiomycosis. FrogLog, 99, 38-39.
Ohmer, M. E., Herbert, S. M., Speare, R., & Bishop, P. J. (2013). Experimental exposure indicates the amphibian chytrid pathogen poses low risk to New Zealand's threatened endemic frogs. Animal Conservation, 16(4), 422-429.
Perfect, A. J., & Bell, B.D. (2005). Assessment of the impact of 1080 on the native frogs Leiopelma archeyi and L. hochstetteri: DOC Research & Development Series 209. Department of Conservation, Wellington. 58 p.
Puig, V. M. (2009). Conservation issues for Hochstetter's frog (Leiopelma hochstetteri): monitoring techniques and chytridiomycosis prevalence in the Auckland region, New Zealand: a thesis presented in partial fulfillment of the requirements for the degree of Master of Science in Conservation Biology at Massey University, Auckland, New Zealand (Doctoral dissertation, Massey University).
Reilly, S., Essner Jr, R., Wren, S., Easton, L., & Bishop, P. J. (2015). Movement patterns in leiopelmatid frogs: insights into the locomotor repertoire of basal anurans. Behavioural Processes, 121, 43-53.
Shaw, E. (2010). Emergence behaviour in a captive population of Hochstetter’s frog (Leiopelma Hochstetteri) at Hamilton Zoo: A trial of recording methods: a report submitted in partial fulfilment of the Post-graduate Diploma in Wildlife Management, Otago, New Zealand (Post-graduate Diploma in Wildlife Management, University of Otago).
Shaw, S. D. (2012). Diseases of New Zealand native frogs (Doctoral dissertation, James Cook University).
Shaw, S. D., Berger, L., Bell, S., Dodd, S., James, T. Y., Skerratt, L. F., Bishop, P. J., & Speare, R. (2014). Baseline cutaneous bacteria of free-living New Zealand native frogs (Leiopelma archeyi and Leiopelma hochstetteri) and implications for their role in defense against the amphibian chytrid (Batrachochytrium dendrobatidis). Journal of Wildlife Diseases, 50(4), 723-732.
Shaw, S. D., Bishop, P. J., Harvey, C., Berger, L., Skerratt, L. F., Callon, K., Watson, M., Potter, J., Jakob-Hoff, R., Goold, M., Kunzmann, N., West, P., & Speare, R. (2012). Fluorosis as a probable factor in metabolic bone disease in captive New Zealand native frogs (Leiopelma species). Journal of Zoo and Wildlife Medicine, 43(3), 549-565.
Shaw, S. D., Bishop, P. J., Skerratt, L. F., Myhre, J., & Speare, R. (2014). Historical trends in frog populations in New Zealand based on public perceptions. New Zealand journal of zoology, 41(1), 10-20.
Shaw, S. D., Skerratt, L. F., Kleinpaste, R., Daglish, L., & Bishop, P. J. (2012). Designing a diet for captive native frogs from the analysis of stomach contents from free-ranging Leiopelma. New Zealand Journal of Zoology, 39(1), 47-56.
Stephenson, E. M. (1951). The anatomy of the head of the New Zealand frog, Leiopelma. The Transactions of the Zoological Society of London, 27(2), 255-305.
Turbott, E. G. (1949). Discovery of the breeding habits of Leiopelma hochstetteri Fitzinger. Records of the Auckland Institute and Museum, 373-376.
van Winkel, D., Baling, M. & Hitchmough, R. (2018). Reptiles and Amphibians of New Zealand: A field guide. Auckland: Auckland University Press, 376 pp.
Whitaker, A. H., & Hardy, G. S. (1985). An unusual frog observation. Journal of the Royal Society of New Zealand, 15(3), 289-290.
Worthy, T. H. (1987). Osteology of Leiopelma (Amphibia: Leiopelmatidae) and descriptions of three new subfossil Leiopelma species. Journal of the Royal Society of New Zealand, 17(3), 201-251.
Worthy, T. H. (1987). Palaeoecological information concerning members of the frog genus Leiopelma: Leiopelmatidae in New Zealand. Journal of the Royal Society of New Zealand, 17(4), 409-420.
Wren, S., Bishop, P. J., Beauchamp, A. J., Bell, B. D., Bell, E., Cisternas, J., Dewhurst, P., Easton, L., Gibson, R., Haigh, A., Tocher, M., & Germano, J. M. (2023). A review of New Zealand native frog translocations: lessons learned and future priorities. New Zealand Journal of Ecology, 47(2), 3538.