- Home
- Herpetofauna Index
- Native
- Woodworthia Maculata
Woodworthia maculata
Raukawa gecko | Moko pāpā
Woodworthia maculata
(Gray, 1845)
Length: SVL up to 89mm, with the tail being equal to or longer than the body length
Weight: up to 14 grams
Description
A widespread, and predominantly ground-dwelling gecko, known for its diverse rock/lichen-like colouration, and patterning. Generally, this is the first species of gecko many of us will come across in the wild (particularly in the North Island) given its terrestrial nature (can be found under things on the ground), and wide distribution (northern South Island, through to eastern Northland (Bay of Islands).
The Raukawa gecko is characterised by its largely grey to brown, rock-like dorsal colouration, which may be broken up by black speckling or irregular lichen-like markings, which primarily consist of a mix of black, white, yellow/orange, and olive green patches. As with many other gecko species these may appear as transverse (blotches or bands), or longitudinal (stripes) markings.
The lateral surfaces of the gecko are much the same as the dorsal surfaces, although, a canthal stripe (between the nostril and eye) may or may not be present on the face.
The ventral (lower) surfaces are usually plain and pale but may have light grey flecking in some individuals.
The Raukawa gecko is also characterised by a pink mouth colouration with a pink/grey tongue.
In comparison to most of our gecko fauna which are fairly arboreal (tree-dwelling) in nature, the Raukawa gecko spends a significant amount of time on the ground, and as such its toe pads appear much thicker than the gracile toes of other genera (e.g., Naultinus, Mokopirirakau, and, Dactylocnemis).
May be confused with the co-occurring forest (Mokopirirakau granulatus), and ngahere geckos (Mokopirirakau "southern North Island"), but in general can be distinguished based on toe shape - thick stubby toes (versus gracile toes), mouth colour - pink (versus orange), and pattern (North Island Mokopirirakau generally have several W-shaped markings running the length of the back, whereas Raukawa geckos generally do not).
May also be confused with the co-occurring Pacific gecko (Dactylocnemis pacificus) in the northern North Island, but can be distinguished based on toe shape (similar to Mokopirirakau), as well as by the position of the rostral scale, with it being separated from the nostril in Raukawa geckos, but in broad contact with them in Pacific geckos.
May be confused with several other Woodworthia species within its range:
Distinguished from the Muriwai gecko (Woodworthia korowai) based on locality - east coast (versus west coast of Auckland).
Distinguished from the goldstripe gecko (Woodworthia chrysosiretica) by the absence of clean unbroken light brown to bright cream dual-striping that carries from the nostril to the end of the tail (present in goldstripe geckos).
Distinguished from the much smaller minimac (Woodworthia "Marlborough mini"), and Kaikōura geckos (Woodworthia "Kaikōura") based on size - >65mm SVL in adults (versus <65mm in adults).
Distinguished from the Kahurangi gecko (Woodworhia "Mt Arthur") based on elevation - with Kahurangi geckos being restricted to the alpine zone.
Life expectancy
Like many of Aotearoa's gecko species, Raukawa geckos are thought to have relatively long life spans. A closely-related species, the Waitaha gecko (Woodworthia cf. brunnea) has been reported living 50+ years in the wild, whilst captive records for Raukawa geckos suggest a life span of at least 37 years.
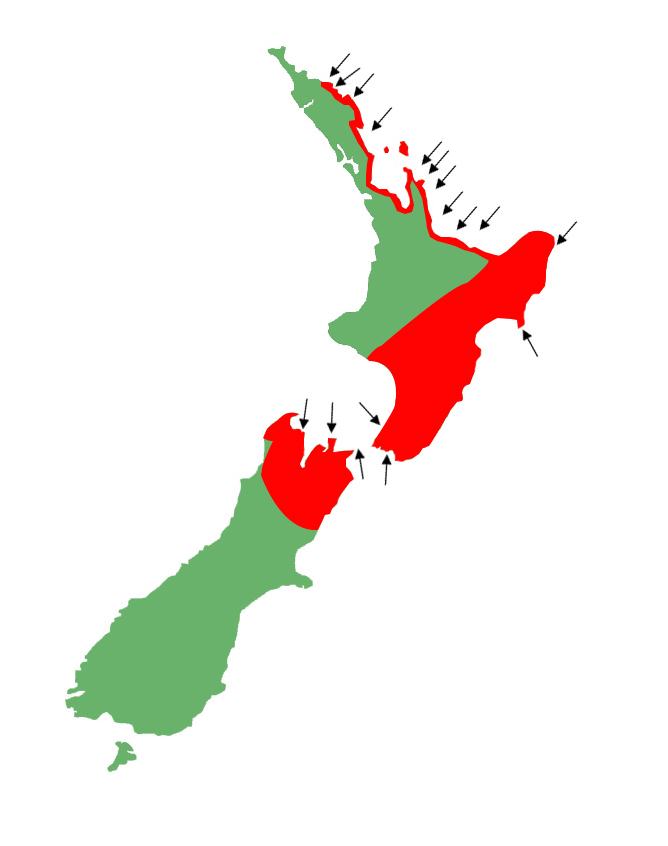
Distribution
Widely distributed throughout the North Island, and the north-western South Island. Strongly associated with coastal habitats throughout their range, but almost exclusively associated with them in the north-eastern North Island, being essentially coastal from the Bay of Islands through to the Bay of Plenty. South of this their distribution pushes further inland, with populations found along the North Islands main fault line from the Urewera's, and Raukumara's, through the Central Plateau, and out to Whanganui. Absent from the North Island's west coast north of the Whanganui River.
In the South Island, Raukawa geckos are confined to the Marlborough, Nelson / Tasman, and West Coast regions.
Ecology and habitat
Raukawa geckos are nocturnal in nature, but are known to cryptically bask at the edges of retreats (often only partially out of cover). As with all members of the Woodworthia genus they are considered terrestrial, and for the most part saxicolous (rock-dwelling) in nature, however, many populations are semi-arboreal in nature, and can often be seen hunting for prey or searching for nectar on the trunks, and foliage of shrubs and trees.
Due to their terrestrial and saxicolous nature, they are often associated with rocky habitats throughout the country, however, they can actually be found in a vast array of habitats, from sandy or rocky coastlines right through to inland beech and broadleaf forests.
Social structure
The Raukawa gecko, as with many of our Woodworthia, is a highly gregarious species which can be found forming large aggregations where suitable refugia are present. However, males have been known to be territorial, particularly during the breeding season.
Breeding biology
Like all of Aotearoa's gecko species, the Raukawa gecko is viviparous, giving birth to one or two live young annually in late Summer. As is the case with many lizard species, mating may seem rather violent with the male repeatedly biting the female around the neck and head area. Sexual maturity is reached between 1.5 to 2 years.
Diet
Raukawa geckos are omnivores. They are primarily insectivorous in nature, but are also known to feed on the nectar and small fruits of several plant species, as well as the honeydew of scale insects when they are seasonally available. Raukawa geckos are often associated with flowering flax, pohutukawa and the honeydew produced by scale insects.
Disease
The diseases and parasites of Aotearoa's reptile fauna have been left largely undocumented, and as such, it is hard to give a clear determination of the full spectrum of these for many species.
The Raukawa gecko, as with many of our other Woodworthia species, is a host for several species of endoparasitic nematodes in the Skrjabinodon genus, as well as at least one strain of Salmonella. The Raukawa gecko is also known to be a host for several ectoparasitic mites including Geckobia haplodactyli, Neotrombicula naultini, Microtrombicula hoplodactyla, and Ophionyssus galeotes
Wild Woodworthia have been found with pseudobuphthalmos (build-up of liquid in the spectacle of the eye) as well as Disecdysis (shedding issues).
In addition to the above, they are known to suffer from several common reptile diseases including black spot fungi, ulcerative dermatitis, mycotic pneumonitis and mycotic septicaemia.
Conservation status
Listed in the most recent threat classification as 'Not Threatened' due to their fairly large and stable nationwide population.
Interesting notes
The specific name ‘maculata’ comes from the Latin word maculō meaning to be spotted. Whilst their common name comes from the reo name for the Cook Strait - Te-Moana-o-Raukawa - which is a stronghold for this species.
Raukawa geckos are one of the most widespread species of Woodworthia, with their distribution including the majority of the North Island, as well as the Cook Strait Islands, Marlborough Sounds and northern South Island. Interestingly at the northern extent of their range - lower Northland to the Bay of Plenty - they are primarily associated with coastal habitats. It has been suggested that this pattern of distribution may be a result of interspecific competition with Pacific geckos (Dactylocnemis pacificus) - a species that shares similarities in its ecology and overlaps within this region (pers.comm Tim Harker).
Raukawa geckos are members of the 'common gecko' complex, a group of closely related species which are regionally distributed throughout New Zealand. Historically, most of these were considered a single highly-variable species - Hoplodactylus maculatus (the so called 'common gecko'). The 'common gecko' has now been separated into over ten different species.
The Raukawa gecko sits within the Northern clade of the Woodworthia complex, with the Korowai gecko being its closest relative within the group.
References
Ainsworth, R. (1985). An evaluation of some island biogeographic theories using lizards and lizard parasites in the Wellington region.
Unpublished master's dissertation. Victoria University: Wellington, New Zealand.
Gill, B., & Whitaker, T. (2007). New Zealand frogs and reptiles. Auckland: David Bateman Limited.
Green, N.D. (2001). Resurvey of a lizard community at Turakirae head, Wellington. Unpublished BSc honours thesis. Victoria University: Wellington, New Zealand.
Hitchmough, R. A. (1979). Lizards observed during a visit to the Cavalli Islands, December 1978 to January 1979. Tane, 25, 119-124.
Jewell, T. (2011). A photographic guide to reptiles and amphibians of New Zealand. Auckland: New Holland Publishers.
Marshall, J.M. (1983). Homing and celestial orientation in two lizards Hoplodactylus maculatus and Leiolopisma nigriplantare. Unpublished BSc honours dissertation. Victoria University: Wellington, New Zealand.
McCallum, J., & Harker, F. R. (1981). Reptiles of Cuvier Island. Tane, 27, 17-22.
McCallum, J., & Hitchmough, R. A. (1982). Lizards of Rakitu (Arid) Island. Tane, 28, 135-136.
Meads, M.J. (1982). A proposed revision of the Naultinus/Heteropholis species complex. In: New Zealand Herpetology. Proceedings of a Symposium held at the Victoria University of Wellington, 29-31 January 1980 (pp. 29-31). Victoria University: Wellington, New Zealand.
Robinson, M. (1985). Sexual cycles of New Zealand lizards, with particular reference to the gecko Hoplodactylus maculatus (Boulenger) (Doctoral dissertation, Victoria University of Wellington).
Towns, D. R. (1971). The lizards of Whale Island. Tane, 17, 61-65.
Twentyman, C. (1999). Diseases in New Zealand reptiles. Surveillance, 26, 3.
van Winkel, D., Baling, M. & Hitchmough, R. (2018). Reptiles and Amphibians of New Zealand: A field guide. Auckland: Auckland University Press, 376 pp.
Whitaker, A.H. (1982). Interim results from a study of Hoplodactylus maculatus (Boulenger) at Turakirae Head, Wellington. In: D.G. Newman (Ed.), New Zealand Herpetology. Proceedings of a Symposium held at the Victoria University of Wellington 29-31 January 1980 (pp. 363-374).
New Zealand Wildlife Service occasional publication No. 2. 495 p.
Raukawa gecko on rock pile (Moutohorā / Whale Island, Bay of Plenty). © Nick Harker